Physics and Technology
Shock waves have many applications in medicine today. Originally used only to break up kidney stones, they are now used in orthopaedic pain therapy or to treat patients with Alzheimer's disease, among other things.
To understand this amazing range of applications, it is worth taking a look at the physical properties of shock waves which lead to their various medical effects:
Focused shock waves
Physical and biological effects of shock waves
Radial pressure waves
Today, extracorporeally generated shock waves and pressure waves are used in the most diverse medical disciplines. Depending on the specific type of application, we refer to Extracorporeal Shock Wave Therapy (ESWT) or – in urinary stone management – to Extracorporeal Shock Wave Lithotripsy (SWL).
This non-invasive therapy procedure dates back to the 1960s, when the idea emerged of generating shock waves extracorporeally and then transmitting them into the body to disintegrate kidney stones and gallstones – without damage to the tissue that the waves pass on their way to the target area.
The first successful stone fragmentation in the human body1,2,3 was performed by Professor Christian Chaussy, M.D., in Munich in February 1980. In the years since, shock waves have been increasingly used for a range of other applications such as the treatment of pseudarthrosis4,5 or of disorders at the site of tendon insertions6. Today, this therapy is employed for many additional conditions, and its potential appears far from being exhausted.
Shock waves were first used on a human patient in early 1980 for the fragmentation of a kidney stone.
Shock waves vs pressure waves
In medical practice, both focused shock waves and radial pressure waves are used. Radial pressure waves are often referred to as radial shock waves, although this is not the correct term as used in physics.
Shock waves and pressure waves differ not only with regard to their mode of generation, but also in terms of the physical parameters generally used and the therapeutic tissue penetration depths achieved.
The following summary provides important background information on the physical principles and technology of shock wave and pressure wave application, and on the differences between them. Should you have any queries after reading this brochure, please do not hesitate to contact us at
Radial pressure waves are often referred to as radial shock waves, although this is not the correct term as used in physics.
Focused shock waves
What are shock waves?
Shock waves are sound waves. They occur in the atmosphere during explosive events, for example during detonations or lightning strikes, or when aeroplanes break through the sound barrier. Shock waves are acoustic pulses characterized by high positive pressure amplitudes and a steep pressure increase compared to the ambient pressure. They are capable of temporarily transmitting energy from the point of generation to remote regions, causing window panes to shatter, for example.
Shock waves propagate explosively and may cause window panes to shatter at great distances.
Shock waves vs ultrasound
Shock waves are similar to ultrasound. But there is a major difference: shock waves have substantially higher pressure amplitudes, which means that steepening effects (Fig. 3) resulting from non-linearities in the propagation medium (water, human tissue) have to be taken into consideration. Another difference is that most ultrasound waves are periodic oscillations with a narrow bandwidth (Fig. 1) whereas shock waves are characterized by a single, mostly positive pressure pulse followed by a comparatively small tensile wave component (negative pressure pulse) (Fig. 2). A pulse of this nature contains frequencies that may range from a few kilohertz to over 10 megahertz.1,7,8
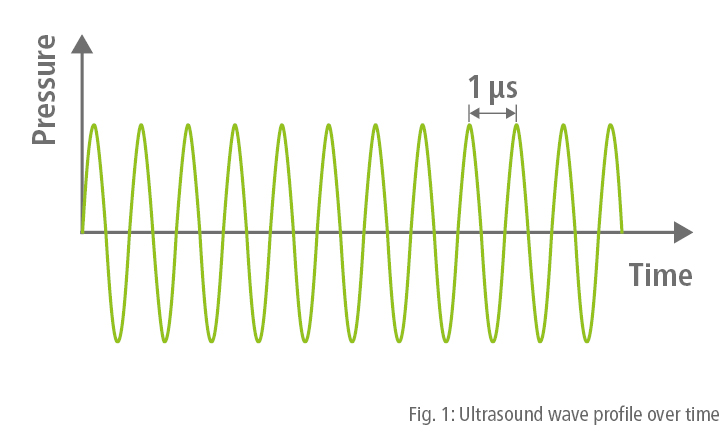


Shock waves are pulses, ultrasound waves are continuous oscillations.
Generation of focused shock waves
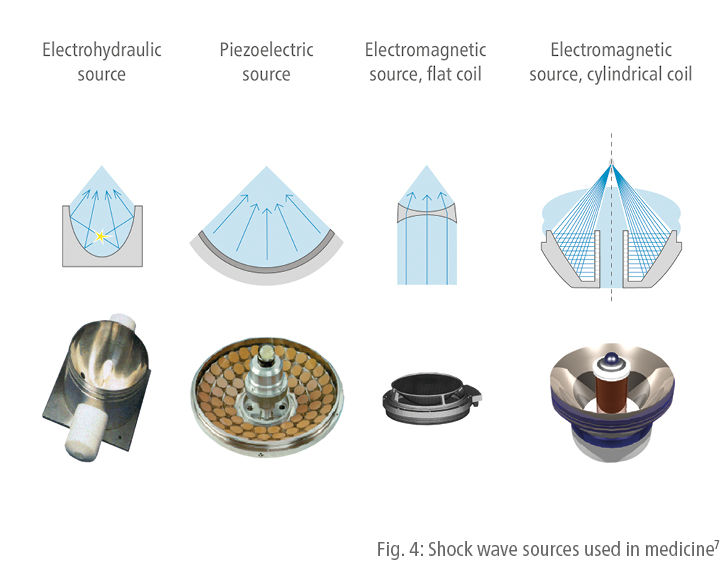
Focused shock waves can be generated by means of electrohydraulic, piezoelectric or electromagnetic shock wave generators (Fig. 4). Electrohydraulic systems produce shock waves directly at the source. Piezoelectric and electromagnetic generators, however, create shock waves as a result of wave steepening and superposition, which means that the wave only forms in the focal zone where the intensity reaches its maximum.
The fact that shock waves produced with different types of generators have differently sized focal zones plays a key role in medical applications. Shock waves generated with the piezo-electric principle have the smallest focus, while those produced with an electrohydraulic source have the largest focus. From this, it follows that the shock wave dose that may be required for a specific treatment partly depends on the type of shock wave system employed.1,8
Example: Electromagnetic shock wave generation
The method of electromagnetic shock wave generation is based on the physical principle of electromagnetic induction. It enables very precise and highly sensitive dosing of the applied shock wave energy. Ideally, the shock wave source used in this case consists of a cylindrical coil combined with a reflector in the form of a rotation paraboloid to focus the shock waves. This provides an accurately defined focal zone, which can be reproduced with maximum precision both axially (in depth) and laterally. Due to the comparatively large aperture of the shock wave source relative to the focus size, the shock wave energy can be introduced into the body over a large coupling area, causing hardly any pain. Most of the shock wave energy is only released in the relatively small focal zone inside the body (Fig. 5).

Shock waves generated with an electromagnetic source cause minimal pain and can be precisely dosed.
Propagation of focused shock waves
Shock waves are acoustic waves. They require a medium such as water or air for propagation. In general, medically used shock waves are generated in water outside the body and then transmitted to the biological tissue. As tissue mainly consists of water, it has very similar sound transmission properties. These properties are described by the acoustic impedance (Z). As a consequence, shock waves are transmitted to the biological tissue without any significant loss. Acoustic impedance is defined as follows:

Acoustic interfaces at which the acoustic properties – i.e. density (ρ) and sound velocity (c) – change, give rise to phenomena familiar from the field of optics such as refraction, reflection, scatter and diffraction, causing the waves to deviate from the straight line of propagation. These effects must be taken into consideration when applying shock waves to the human body. This is crucial to ensure that the applied energy is effective in the treatment zone.
Shock waves are reflected and refracted at acoustic interfaces (as happens when light hits a mirror). The greater the difference between the acoustic impedances of two media, the stronger this effect will be.
For this reason, the first device for kidney stone fragmentation required the patient to be submerged in a water-filled tub. Today’s systems involve »dry« coupling, which means that the water bath is connected to the body via a flexible coupling membrane. Trapped air in between is eliminated by means of coupling gel or a thin water film.
Trapped air or air bubbles between the shock wave source and the body significantly diminish the effectiveness of shock waves.
In addition to this, it is important that no gas-filled organs (lungs) or large bone structures are located on the shock wave propagation path. They would act as obstacles to the transmission of shock waves to the target area and thus inhibit the desired therapeutic effect. Moreover, the release of shock wave energy at gas-filled organs would cause damage to the tissue (contraindication).
We also need to assume that different types of soft tissue (skin, fat, muscles, tendons, etc.) have inhomogeneous acoustic properties and that they do have interfaces. However, the differences in the acoustic properties are considerably less pronounced than at the interfaces between water and air. In addition to absorption and reflection, refraction effects occur here which may lead to difficult-to-control deviations from the straight line of propagation of shock waves inside the body.
Shock wave parameters / Shock wave measurement / Shock wave pressure
Measurements with pressure sensors are the preferred method of identifying the characteristics of shock waves.8 Shock waves used in medicine (Fig. 2) typically have p+ peak pressures of about 10 to 100 megapascals (MPa), which is equivalent to about 100 to 1000 times the atmospheric pressure. Depending on the shock wave generation method used, rise times are very short at around 10 to 100 nanoseconds (ns). The pulse duration is approximately 1000 nanoseconds (1 µs) (and hence much shorter than that of the medical pressure waves, see Fig. 15). Another characteristic of shock waves is the relatively low p- tensile wave component, which is around 10% of the p+ peak pressure.
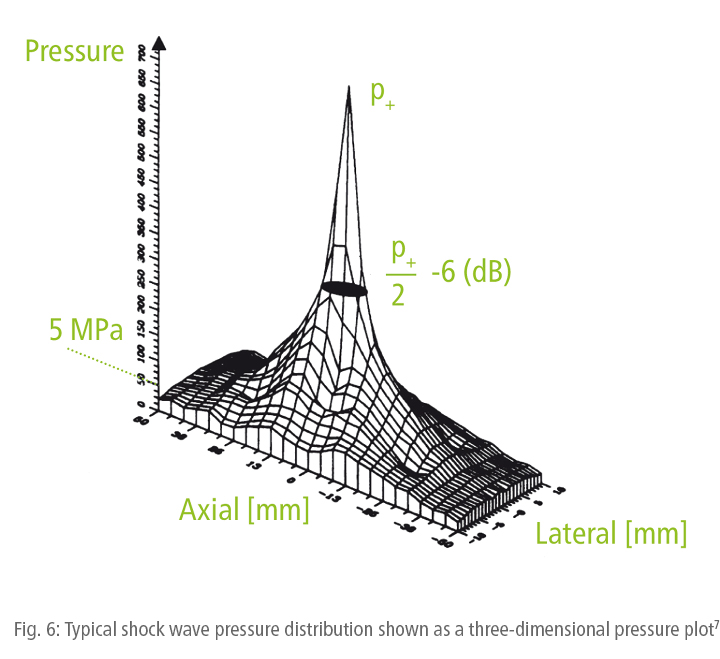
If the p+ peak pressure values measured in the focal zone are plotted in a three-dimensional graph (coaxially to the shock wave propagation path and laterally, i.e. perpendicularly, to this direction), the typical pressure distribution obtained is as shown in Fig. 6. Evidently, the shock wave field does not have clear boundaries, but the shape of a mountain with a peak in the centre and more or less steep slopes. This is referred to as the three-dimensional pressure distribution model. The shape and height of this model may differ, depending on which type of shock wave system is used.
Shock wave focus
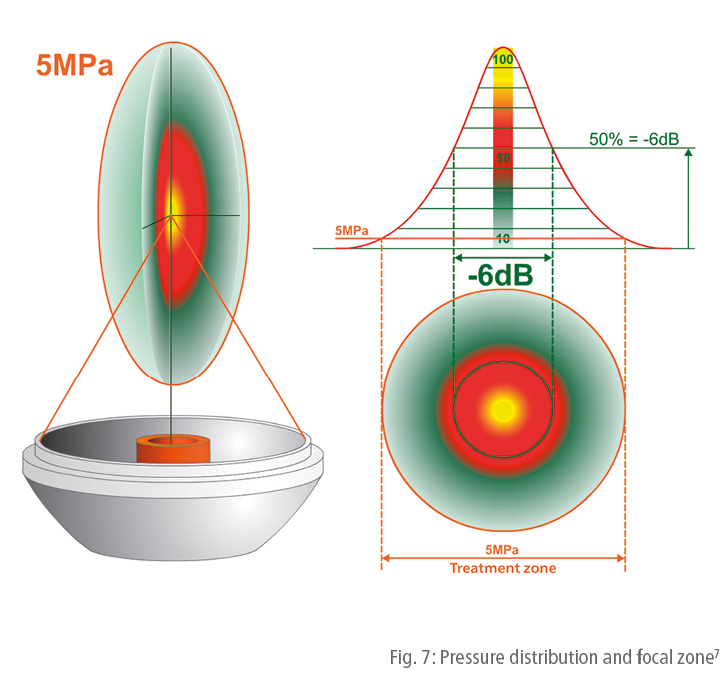
The shock wave focus is defined as the area within the mountain-like pressure distribution model in which the pressure is equal to or higher than 50% of the peak pressure (Fig. 6 and 7). This area is also referred to as the -6dB focal zone or described using the acronym FWHM (full width at half maximum).
5 MPa treatment zone
The area in which the shock wave produces its biological effects can only be gauged when taking into consideration the specific energy level. In other words: the shock wave treatment area inside the body is not identical with the size of the -6dB focal zone. It can be larger or smaller. This is why an additional parameter has been defined, which is more closely related to the therapeutic effectiveness of shock waves and which is not based on relative values (relationship to the peak pressure in the centre), but rather on an absolute quantity, specifically the 5 MPa pressure (50 bar). Consequently, the 5 MPa focus has been defined as the spatial zone in which the shock wave pressure is equal to or above 5 MPa. This definition is based on the assumption that a certain minimum pressure threshold exists below which shock waves have no or only minimal therapeutic effectiveness.
To date, there is no scientific evidence to support the 5 MPa value. However, the above definition also reflects changes in the treatment zone resulting from changes in the selected energy level. Different therapy zones and their changes with different energy levels are shown in schematic form in Fig. 8. In contrast to the treatment zone, the -6dB focal zone basically remains the same even if the energy settings change.
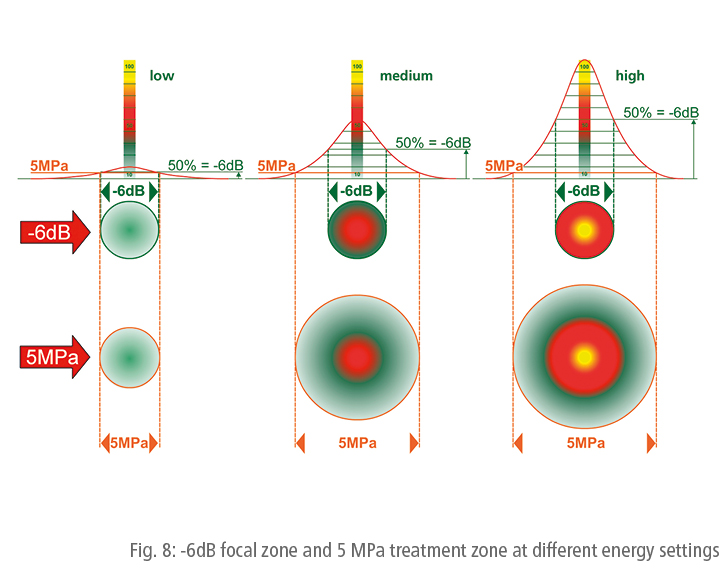
The focal zone is the area of maximum energy intensity. Its size is basically independent of the selected energy level. By contrast, the size of the treatment zone depends on the selected energy level and is generally larger than the focal zone.
Energy (E)
Shock wave energy is a key parameter5 in medical shock wave application even if, today, greater importance is being attributed to energy flux density. It can be assumed that shock waves only have an effect on tissue when certain energy thresholds are exceeded. The energy is determined by integration from the pressure curve plotted over time p(t). It is proportional to the surface area (A) and inversely proportional to the acoustic impedance (Z):

A distinction is made as to whether integrating the pressure over time only includes the positive pressure components (E+) or whether it also incorporates the negative (tensile) components (Etotal). The total energy is usually stated as E (without index). The acoustic energy of a shock wave pulse is given in millijoules (mJ). As a rule, several hundred or thousand shock wave pulses are applied per treatment session. This means that the total amount of energy applied is calculated through multiplication by the number of pulses.1,8
Energy flux density (ED)
The therapeutic effectiveness of shock waves depends on whether the shock wave energy is distributed over a large area or focused on a locally confined treatment zone (focal zone). A measure of the energy concentration is obtained by calculating the energy per area (E/A):

The energy flux density ED is given in millijoules per square millimetre (mJ/mm2). Here again, a distinction is made between (on the one hand) integration over the positive part of the pressure curve alone and (on the other) inclusion of the negative component. If specified without index (ED), the pressure curve is usually considered to include the negative (tensile) component (total energy flux density).
The first shock wave systems were equipped with an electrohydraulic shock wave generator. Unlike today, the energy levels were not given in mJ/mm2, but were specified as voltage values (kV).
Momentum
A fact that has received little attention thus far is that shock waves have momentum. As with its energy, a shock wave’s momentum is defined in terms of the integration of pressure over time. Unlike a shock wave’s energy, however, its momentum is not squared prior to being integrated. Its sign is thus retained and the momentum’s vectorial nature means there is both momentum due to the positive pressure component in the shock wave’s direction of propagation, and lesser momentum in the backwards direction generated by the negative tensile wave component.
The asymmetrical pulse form of the shock wave ensures that both successive momentums cannot compensate for each other, and a reciprocal effect with high pressure and low tension is generated. This is not the case with continuous ultrasound: here the alternating tension and pressure phases largely cancel each other out, so that the resulting momentum is relatively small in magnitude.



Shock wave momentum is of crucial importance both for high-energy stone fragmentation and for the low-energy medico-biological stimulating effect.34
The momentum of a shock wave is particularly important since it is the means by which forces are exerted upon the matter. If a shock wave is propagated in, say, biological tissue, interference to this wave will be only minimal, provided it does not strike tissue interfaces at which there are abrupt changes in tissue density ρ and/or the velocity of sound c within this medium, and hence in the acoustic impedance Z = ρ x c. At such interfaces, the momentum of the wave is divided by reflection into two parts: one that passes right through the interface, and another that is reflected back from it. The relative proportions of the transmitted and the reflected wave are determined by the reflection coefficient.

This means that the greater part of the wave is reflected at hard interfaces like stones, and the smaller part at soft interfaces including tendons and muscle fibres. Directly related to this are forces which are exerted on the interfaces, and can be used both to fragment material that is amenable to breakage (such as calculi) and to stimulate elastic materials including soft body tissue.
The physical foundation for the release of force when reflection takes place was recognized as long ago as 1687 by Isaac Newton, who stated that any change in momentum necessarily involves the action of force.
Generally, the following holds (after Newton):

Specifically: a body of mass m, moving at velocity v, possesses momentum P = mv. If this body is accelerated or decelerated, its velocity changes. This involves the action of a force F = m(dv/dt). Depending on the direction of this force, it can be termed an accelerating or decelerating force.
Physical and biological effects of shock waves
Direct effects on interfaces
The selective mechanism of action of shock waves on contact with different tissues and material interfaces went unacknowledged for a long time. It does, however, form the basis for various medical applications. And it explains why shock waves can pass through many types of tissue without causing appreciable damage, and why a therapeutic release of force that may even fragment calculi is selectively observable at interfaces. Therefore, the effect of shock waves in medicine can be attributed to the physical mechanism of momentum transfer and the action of force that this causes.
It is not only in near-surface tissue that shock waves can exert forces. Due to the high-frequency components within the megahertz range, and the associated short pulse lengths of only a few millimetres, shock waves can be focused on deeper regions within the body. Here, the mechanism of momentum transfer means they can specifically generate longer stimulation pulses in the physiologically effective millisecond range.
The forces which, subject to these physical laws, arise at tissue interfaces during momentum transfer by means of transmission and selective reflection, are able to produce slight movements of these interfaces. These movements ensure that layers of cells are stretched and deformed, briefly becoming permeable to ions and certain molecules. This mechanism is referred to as mechanotransduction, which is currently held to be the key factor in numerous mechanisms of action involved in medical applications of shock waves. This is because mechanotransduction offers an explanation as to why a number of biochemical substances – such as nitric oxides (NO), growth factors and substance P – are released.18,21 And shock waves have another important characteristic. As with ultrasound, the chief pressure-related processes take place in the microsecond range, i.e. much too rapidly for most physiological processes. By contrast, the physiology of mechanotransduction requires force effects of much longer duration, namely in the millisecond range. Exchange of biochemical substances through briefly opened pores in stretched membranes is possible within this timeframe. Shock wave momentum transfer when reflection takes place at interfaces ensures that the masses involved are accelerated and carry out slight movements within the duration of milliseconds.

The characteristics of shock waves and ultrasound waves are different. Ultrasound exerts a high-frequency alternating load on the tissue in the frequency range of several megahertz; at high amplitudes, this leads to heating, tissue tears and cavitation.9,10 The effect of shock waves is determined by, among other factors, a forward-directed force effect (in the direction of shock wave propagation), which causes momentum transfer to the interface (see explanations above).
This force effect can be increased to such an extent that even kidney stones can be destroyed.2,3 In general, force effects of this nature occur at interfaces characterized by discontinuities in the acoustic impedance, but hardly ever in homogeneous media (tissue, water).11 As a result, shock waves are the ideal means for creating effects in deep tissue without causing damage to the tissue that the waves pass on their way to the target area.
However, even less distinct interfaces within soft tissue structures experience a minor force effect from the application of shock waves. Depending on shock wave intensity (Fig. 11), mechanical destruction of cells, membranes and bone trabeculae11 may, for example, occur as may cellular stimulation through reversible deformation of the cell membrane12. The outcomes that can be achieved in this manner are twofold: the destruction of structures amenable to breakage (kidney stones) and the irritation and stimulation of tissue structures with consequential healing processes. This phenomenon is evident in orthopaedic applications, for example.36 Focusing of shock waves allows the desired effect to be confined to the target area, so that side effects outside the treatment zone can be reduced or even completely avoided.
Shock wave focusing enables targeted treatment of a confined area.
It has been shown that, in most cases, shock wave therapy results in increased blood circulation and enhanced metabolic activity, leading to the onset of the healing process.
Shock waves also induce a variety of biological reactions resulting from the shear and pressure forces they produce (see explanations above). This mechanism of action is referred to as mechanotransduction. As demonstrated by scientific evidence, it is responsible for the following effects:
- Increase in cell permeability15
- Stimulation of microcirculation (blood, lymph)16,17
- Release of substance P18
- Reduction of non-myelinated nerve fibres19
- Release of nitric oxide (NO), which leads to vasodilation, increased metabolic activity and angiogenesis and has an anti-inflammatory effect20,21
- Antibacterial effect22
- Release of growth hormones (blood vessels, epithelium, bones, collagen, etc.)20,23,24,25
- Stimulation of stem cells26,27
- Stimulation of neurons (neural spiking)18,33,35

Indirect effects – cavitation
In addition to the direct force effect of shock waves on interfaces, a phenomenon referred to as cavitation occurs in specific media such as water and, to a certain extent, tissue.9
Cavitation bubbles occur directly after the pressure/tension alternating load of the shock waves has passed through the medium. The majority of the bubbles grow for about 100 microseconds after the waves have passed, and then violently collapse while emitting secondary spherical shock waves. When close to interfaces, cavitation bubbles can no longer collapse without being disturbed. The medium flowing back into the bubble (e.g. water or body fluid) can no longer flow unhindered. Therefore, the bubble collapses asymmetrically while developing a microjet.13 This microjet is directed at the interface at a velocity of several hundred metres per second (Fig. 12).
The microjets contain a high amount of energy and penetrative power so that they are able to erode the hard interfaces of stones. As the shock waves pass through the medium, gas dissolved in the blood or tissue is released and forms bubbles. This phenomenon is referred to as soft cavitation. The cavitation bubbles formed in this manner may tear open blood vessels and cells. This causes micro-bleeding or membrane perforation. Cavitation is not limited to the focal zone alone, but it is particularly pronounced there.1,8,14

Targeted application of focused shock waves
Targeted application of shock waves requires the focal zone of the shock wave system to be directed at the body region requiring treatment. When treating stones (lithotripsy), bones and specific tissue structures, X-ray or ultrasound systems can be used for this purpose. In pain therapy, effective communication with the patient is necessary to identify the point of maximum pain. This »biofeedback« method helps to localize many superficial and deep treatment points.
Radial pressure waves
What are radial pressure waves?
In addition to focused shock waves, modern medicine also uses radial pressure waves. Physicist Sir Isaac Newton established his famous law of »action and reaction« as early as 1687. The method of action of a ballistic pressure wave system is based precisely on the principle of linear momentum deduced from Newton’s law.

Pressure wave therapy is based on the law of »action and reaction« established by physicist Sir Isaac Newton in 1687.
Mechanical energy in the form of an acoustic pressure wave is transmitted to the body tissue and, consequently, to the pain region by means of specially shaped transmitters. Introduced in the late 1990s, ballistically generated radial pressure waves are a lower-cost alternative to shock waves, especially in the treatment of musculoskeletal disorders.
Many conditions that indicate the use of radial pressure waves, and the successful therapeutic outcomes achieved, are very similar to those associated with focused shock waves.28 In medical practice, the term »Radial Shock Wave Therapy« (RSWT) is therefore commonly used.
Radial pressure waves are often referred to as radial shock waves, although this is not the correct term as used in physics.
Physicists would say it is incorrect to call radial pressure waves »shock waves«. The pulse length of radial pressure waves is much longer than that of shock waves. Pressure waves have pulse lengths of between 0.15 and 1.5 m. By contrast, the pulse length of shock waves is only about 1.5 mm. This explains why shock waves, unlike pressure waves, can be focused.29
In practice, radial pressure waves are commonly referred to as radial shock waves. However, they have a significantly longer pulse duration than focused shock waves.
Generation of radial pressure waves
Pressure waves are generated by the collision of solid bodies (Fig. 14). First of all, a projectile is accelerated using, for instance, compressed air (as in an air rifle) to a speed of several metres per second (approximately 5 to 25 m/s, i.e. far below the sound velocity in water of about 1500 m/s) and then abruptly slowed down by hitting an impact body (transmitter). The elastically suspended impact body is brought into direct contact with the patient's skin above the area to be treated, preferably using ultrasound coupling gel. When the projectile strikes the impact body, some of its kinetic energy is transmitted to the impact body. The impact body then performs a translational movement over a short distance (typically < 1 mm) at a significantly slower speed (typically < 1 m/s) until the coupled tissue or the handpiece stops the impact body movement. The motion of the impact body is transmitted to the tissue at the point of contact, from where it propagates divergently in the form of a radial pressure wave.

The duration of the pressure pulse (Fig. 15) is determined by the translational movement of the impact body and is typically about 0.2 to 5 milliseconds (ms) in tissue. This means that the pressure pulses applied to the tissue are longer by a factor of 1000 than those of shock waves. Typical peak pressures of radial pressure waves are about 0.1 to 1 MPa, i.e. significantly lower – by a factor of 100 – than those of shock waves.1,8
The collision of the projectile with the impact body also generates a higher-frequency acoustic wave (solid-borne sound) in the impact body. Owing to the great difference between the two acoustic impedances (metal, water), only a minimal portion (about 10%) of this oscillation energy is transmitted to the tissue or water. The energy contained in the high-frequency acoustic oscillation is significantly smaller than the energy of the low-frequency pressure pulse described above.30

Propagation of pressure waves
Pressure waves as described here originate from the application point of the impact body and travel radially into the adjacent tissue.29 The energy density of the induced pressure wave rapidly drops with increasing distance from the application point (by a proportion of 1/r2). This means that the strongest effect is at the application point of the impact body, i.e. at the skin surface (Fig. 16).

The therapeutic effectiveness of radial pressure waves reaches a depth of 3 to 4 cm, but it is strongest at the skin surface.
Pressure wave parameters / Pressure wave measurement
Due to the significantly longer pulse duration and low-pressure amplitude of pressure waves compared to shock waves, pressure measurements in water (as commonly performed for shock waves) would not provide conclusive results. More accurate information can be obtained by measuring the excursion of the impact body (Fig. 17) and the force transmitted to a viscoelastic tissue phantom. However, since these parameters strongly depend on the type of impact body (transmitter) used, the intensity parameter commonly given is the pressure that drives and accelerates the projectile.

Physical and biological effects of pressure waves
Radial pressure waves generate oscillations in tissue which lead to improved microcirculation and increased metabolic activity.31 Despite the multitude of successful treatment outcomes, hardly any scientific research has been conducted so far to investigate the precise biological effects of radial pressure waves.
Interestingly, despite the physical differences and the resulting different application areas (superficial or deep target areas), the stimulation effects and therapeutic mechanisms seem to present certain similarities. Radial pressure waves are ideal for the treatment of superficial pain, for example. In the therapy of myofascial pain syndromes, radial pressure waves are indispensable for smoothing muscles and/or fascia before or after focused shock wave application.
Similarities in the effects of focused shock waves and radial pressure waves can beexplained in terms of mechanotransduction as the underlying mechanism in both cases. Whereas, when focused shock waves are applied, their short wavelength makes it possible for the effects of the stimulation pulse to be specifically exerted either superficially or at depth, with radial pressure waves this can – since they cannot be focused – be achieved only at the surface, with the effect at depth diminishing radially.
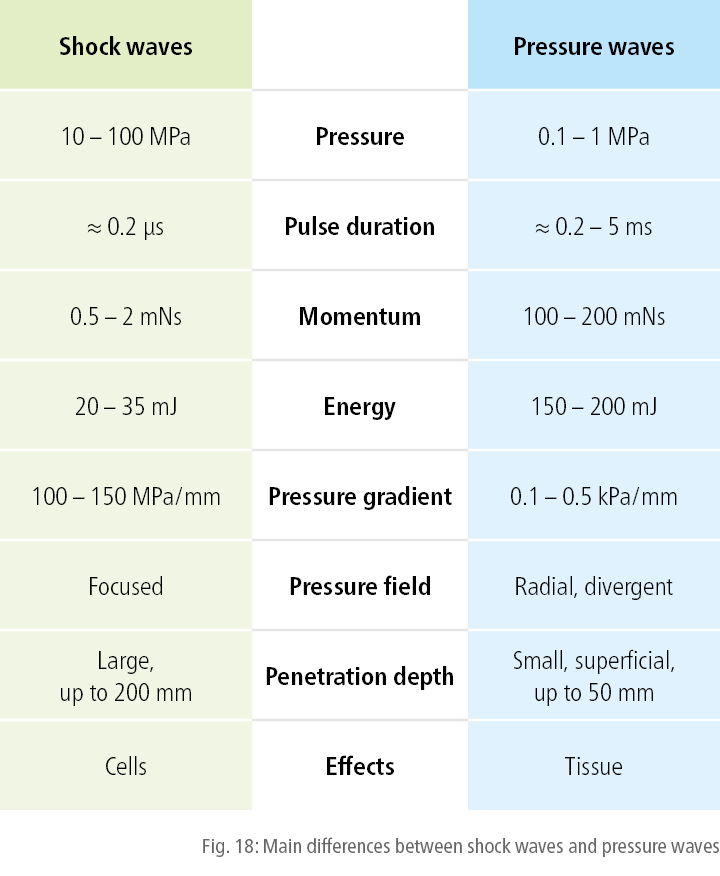
Shock waves vs pressure waves
Shock waves and pressure waves differ not only with regard to their physical properties and mode of generation, but also in terms of the magnitude of the standard parameters used and the therapeutic tissue penetration depths achieved. The main differences are summarized in Fig. 18.
Local painful spots, chronic enthesopathies and deep trigger points should be treated with focused shock waves.32
Similarities in the effects of focused shock waves and radial pressure waves can beexplained in terms of mechanotransduction as the underlying mechanism in both cases. Whereas, when focused shock waves are applied, their short wavelength makes it possible for the effects of the stimulation pulse to be specifically exerted either superficially or at depth, with radial pressure waves this can – since they cannot be focused – be achieved only at the surface, with the effect at depth diminishing radially.
Shock waves vs pressure waves
Shock waves and pressure waves differ not only with regard to their physical properties and mode of generation, but also in terms of the magnitude of the standard parameters used and the therapeutic tissue penetration depths achieved. The main differences are summarized in Fig. 18.
Local painful spots, chronic enthesopathies and deep trigger points should be treated with focused shock waves.32
1 Wess, O.: Physikalische Grundlagen der extrakorporalen Stoswellentherapie. Journal für Mineralstoffwechsel, 11(4), 7 – 18, 2004.
2 Chaussy, C. et al.: Extracorporeally induced destruction of kidney stones by shock waves. The Lancet, 316(8207), 1265 – 1268, 1980.
3 Chaussy, C. et al.: First clinical experiences with extracorporeally induced destruction of kidney stones by shock waves. The Journal of Urology, 127(3), 417 – 420, 1982.
4 Valchanov, V. et al.: High energy shock waves in the treatment of delayed and nonunion of fractures. International Orthopaedics, 15(3), 181 – 184, 1991.
5 Schaden, W. et al.: Extracorporeal shock wave therapy (ESWT) in 37 patients with non-union or delayed osseous union in diaphyseal fractures. In: Chaussy, C. et al. (eds.): High Energy Shock Waves in Medicine, Georg Thieme Verlag, Stuttgart, 1997.
6 Dahmen, G. P. et al.: Die Extrakorporale Stosswellentherapie in der Orthopädie – Empfehlungen zu Indikationen und Techniken. In: Chaussy, C. et al. (eds.): Die Stosswelle – Forschung und Klinik. Attempto Verlag, Tübingen, 1995.
7 Wess, O.: Physics and technology of shock wave and pressure wave therapy. ISMST Newsletter 2(1), 2 – 12, 2006.
8 Wess, O. et al.: Working group technical developments – consensus report. In: Chaussy, C. et al. (eds.): High Energy Shock Waves in Medicine. Georg Thieme Verlag, Stuttgart, 1997.
9 Church, C.: A theoretical study of cavitation generated by an extracorporeal shock wave lithotripter. The Journal of the Acoustical Society of America, 86(1), 215 – 227, 1989.
10 Church, C.: The risk of exposure to diagnostic ultrasound in postnatal subjects. Journal of Ultrasound in Medicine, 27(4), 565 – 592, 2008.
11 Delius, M. et al.: Biological effects of shock waves: in vivo effect of high energy pulses on rabbit bone. Ultrasound in medicine and biology, 21(9), 1219 – 1225, 1995.
12 Forssman, B. et al.: Stosswellen in der Medizin, Medizin in unserer Zeit. 4: 10, 1980.
13 Crum, L. A.: Cavitation on microjets as a contributory mechanism for renal calculi disintegration in ESWL. The Journal of Urology, 140(6), 1587 – 1590, 1988.
14 Coleman, A. J. et al.: Acoustic cavitation generated by an extracorporeal shockwave lithotripter. Ultrasound in medicine and biology, 13(2), 69 – 76, 1987.
15 Byron, C. R. et al.: Effects of radial shock waves on membrane permeability and viability of chondrocytes and structure of articular cartilage in equine cartilage explants. American Journal of Veterinary Research, 66(10), 1757 – 1763, 2005.
16 Kisch, T. et al.: Repetitive shock wave therapy improves muscular microcirculation. Journal of Surgical Research, 201(2), 440 – 445, 2016.
17 Goertz, O. et al.: Short-term effects of extracorporeal shock waves on microcirculation. Journal of Surgical Research, 194(1), 304 – 311, 2015.
18 Maier, M. et al.: Substance P and prostaglandin E2 release after shock wave application to the rabbit femur. Clinical Orthopaedics and Related Research, (406), 237 – 245, 2003.
19 Klonschinski, T. et al.: Application of local anesthesia inhibits effects of low-energy extracorporeal shock wave treatment (ESWT) on nociceptors. Pain Medicine, 12(10), 1532 – 1537, 2011.
20 Nishida, T. et al.: Extracorporeal cardiac shock wave therapy markedly ameliorates ischemia-induced myocardial dysfunction in pigs in vivo. Circulation, 110(19), 3055 – 3061, 2004.
21 Mariotto, S. et al.: Extracorporeal shock waves: From lithotripsy to anti-inflammatory action by NO production. Nitric Oxide, 12(2), 89 – 96, 2005.
22 Horn, C. et al.: The effect of antibacterial acting extracorporeal shockwaves on bacterial cell integrity. Medical Science Monitor, 15(12), 364 – 369, 2009.
23 Chao, Y.-H. et al.: Effects of shock waves on tenocyte proliferation and extracellular matrix metabolism. Ultrasound in medicine and biology, 34(5), 841 – 852, 2008.
24 Christ, Ch. et al.: Improvement in skin elasticity in the treatment of cellulite and connective tissue weakness by means of extracorporeal pulse activation therapy. Aesthetic Surgery Journal, 28(5), 538 – 544, 2008.
25 Gollwitzer, H. et al.: Radial extracorporeal shock wave therapy (rESWT) induces new bone formation in vivo: results of an animal study in rabbits. Ultrasound in medicine and biology, 39(1), 126 – 133, 2013.
26 Schuh, C. M. et al.: In vitro extracorporeal shock wave treatment enhances stemness and preserve multipotency of rat and human adipose-derived stem cells. Cytotherapy, 16(12), 1666 – 1678, 2014.
27 Raabe, O. et al.: Effect of extracorporeal shock wave on proliferation and differentiation of equine adipose tissue-derived mesenchymal stem cells in vitro. American Journal of Stem Cells, 2(1), 62 – 73, 2013.
28 Auersperg, V. et al.: DIGEST-Leitlinien zur Extrakorporalen Stosswellentherapie, www.digest-ev.de, 2012.
29 Cleveland, R. O. et al.: Acoustic field of a ballistic shock wave therapy device. Ultrasound in medicine and biology, 33(8), 1327 – 1335, 2007.
30 Uberle, F. et al.: Ballistic pain therapy devices: measurement of pressure pulse parameters. Biomedical Engineering/ Biomedizinische Technik, 57 (SI-1 Track-H), 700 – 703, 2012.
31 Grecco, M. V. et al.: One-year treatment follow-up of plantar fasciitis: radial shockwaves vs. conventional physiotherapy. Clinics, 68(8),1089 –1095, 2013.
32 Gleitz, M.: Die Bedeutung der Trigger-Stoßwellentherapie in der Behandlung pseudoradikularer Cervicobrachialgien. Abstracts 53. Jahrestagung der Vereinigung Süddeutscher Orthopäden e.V., 2005.
33 Wess, O.: A neural model for chronic pain and pain relief by extracorporeal shock wave treatment. Urological Research, 2008; 36(6), 327 – 334, 2008.
34 Wess, O. et al.: Fragmentation of brittle material by shock wave lithotripsy. Momentum transfer and inertia: a novel view on fragmentation mechanisms. Urolithiasis, 48(2), 137 – 149, 2020.
35 Beisteiner, R. et al.: Transcranial Pulse Stimulation with Ultrasound in Alzheimer’s Disease – A New Navigated Focal Brain Therapy. Advanced Science, 7(3):1902583, 2019. doi: 10.1002/advs.201902583.
36 Reilly, J. M. et al.: Effect of Shockwave Treatment for Management of Upper and Lower Extremity Musculoskeletal Conditions: A Narrative Review. PM&R, 10(12), 1385 – 1403, 2018.