Jo J. et al., 2025: Investigation of shockwave treatment for disruption of bacterial biofilm on tubular structure.
Jihye Jo 1 2, Sarah Stadler 3, Peter Costandi 3, Thomas Hasenberg 3, Hyun Wook Kang 4 5 6
1Industry 4.0 Convergence Bionics Engineering, Department of Biomedical Engineering, Pukyong National University, Busan, 48513, Republic of Korea.
2Research Center for Marine-Integrated Biomedical Technology, The National Key Research Institutes in Universities, Pukyong National University, Busan, 48513, Republic of Korea.
3Johnson & Johnson Med Tech, New Brunswick, NJ, USA.
4Industry 4.0 Convergence Bionics Engineering, Department of Biomedical Engineering, Pukyong National University, Busan, 48513, Republic of Korea.
5Research Center for Marine-Integrated Biomedical Technology, The National Key Research Institutes in Universities, Pukyong National University, Busan, 48513, Republic of Korea.
6Major of Biomedical Engineering, Division of Smart Healthcare and Digital Healthcare Research Center, College of Information Technology and Convergence, Pukyong National University, 45 Yongso-ro Nam-gu, Busan, 48513, Republic of Korea.
Abstract
Biofilms, which are structures formed by microorganisms, are protected by extracellular polymeric substances (EPS) secreted by bacteria against external threats, including antibiotics. The current study aims to assess the effects of shockwave treatment combined with antibiotic therapy on Pseudomonas aeruginosa biofilms in tubular structures in vitro. The biofilms were formed on the inner surfaces of silicone tubes for three days under dynamic conditions. The biofilms were treated with shockwave treatment (120 pulses at 2 Hz), followed by exposure to 4 µg/ml ciprofloxacin for 6 h. Bacterial viability was assessed using colony-forming unit (CFU) and confocal laser scanning microscopy (CLSM) with SYTO9/PI staining, while biofilm detachment was evaluated via crystal violet (CV) staining and scanning electron microscopy (SEM). According to the SEM and CFU analysis, the shockwave and antibiotic-combined treatment significantly detached the biofilm, removing up to 97.5% of the surface area and decreased bacterial viability by 40%, compared to untreated control biofilms. The CV staining showed a significant reduction in biofilm biomass to an OD600 of 0.14. The CLSM analysis revealed a dead bacteria proportion of 67%. In conclusion, the shockwave treatment combined with antibiotics could effectively degrade the biofilms in tubular structures and enhance antibiotic efficacy.
Sci Rep. 2025 Oct 15;15(1):36041. doi: 10.1038/s41598-025-19955-y. PMID: 41094089;
PMCID: PMC12528757


Comments 1
The study investigates the effectiveness of shockwave treatment combined with antibiotic therapy in disrupting bacterial biofilms formed by pseudomonas aeruginosa in tubular structures. Biofilms, which are protective layers formed by bacteria, pose significant challenges in treating infections, particularly in clinical settings where medical devices are involved.
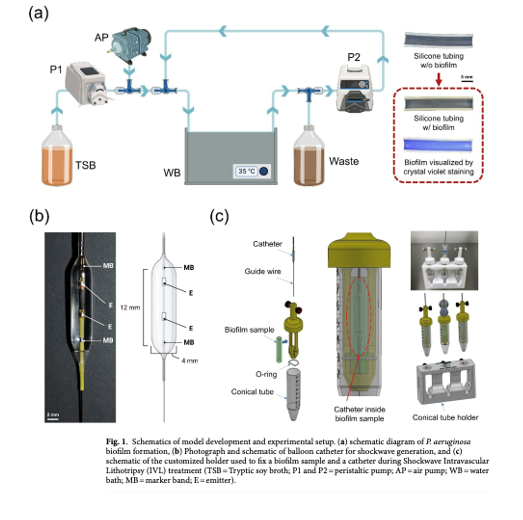
In this in vitro study, biofilms were cultivated on silicone tubes and subjected to shockwave treatment using an intravascular system (120 pulses at 2 Hz) followed by exposure to ciprofloxacin (4 µg/ml) for 6 hours.
Various techniques, including colony-forming unit (CFU) analysis, crystal violet (CV) staining, scanning electron microscopy (SEM), and confocal laser scanning microscopy (CLSM), were used to assess biofilm detachment and bacterial viability. The results indicated that the combined treatment effectively detached biofilms (up to 97.5% surface area reduction) and reduced bacterial viability by 40%, demonstrating a significant enhancement in antibiotic efficacy due to shockwave treatment.
The study concludes that shockwave treatment can effectively degrade biofilms in tubular structures and improve the performance of antibiotics against embedded bacteria.
Strengths of the study
1. Innovative Approach: The combination of shockwave therapy and antibiotics represents a novel strategy for tackling biofilm-related infections, which are notoriously difficult to treat.
2. Comprehensive Methodology: The use of multiple assessment techniques (CFU, CV, SEM, CLSM) provides a robust analysis of both biofilm integrity and bacterial viability.
3. Quantitative Findings: The study presents quantitative data showing the effectiveness of the combined treatment, which strengthens the conclusions drawn about the treatment's efficacy.
4. Clinical Relevance: The focus on biofilms in tubular structures is particularly relevant to clinical challenges associated with medical devices, highlighting the potential for practical applications.
Limitations of the study
1. In Vitro Nature: The study is conducted in vitro, which may not fully replicate the complexities of in vivo environments, including host responses and interactions with other microbial species.
2. Limited Bacterial Strain Testing: The study only examines pseudomonas aeruginosa, which may limit the generalizability of the findings. Different bacterial species may exhibit varied responses to shockwave treatment. However, it has to be mentioned, that other studies have examined similar effect using strains of E. coli.
3. Absence of Additional Control Groups: The study lacks a control group that combines sham treatment with ciprofloxacin, making it difficult to isolate the effects of catheter insertion from those of the shockwave treatment and the antibiotic.
4. Unclear Mechanisms: While the study suggests that shockwave treatment disrupts biofilm integrity and enhances antibiotic penetration, the specific biochemical changes within the biofilm matrix remain unexplored.
Conclusion
The study presents promising findings on the combined use of shockwave treatment and antibiotics to effectively disrupt bacterial biofilms in tubular structures, potentially offering a new therapeutic avenue for managing persistent infections. However, further research, particularly in vivo studies and exploration of various bacterial strains, is needed to validate the findings and optimize treatment protocols for clinical application.
Jens Rassweiler