Tikkinen KAO. et al., 2025: A Multicenter Randomized Controlled Trial of Antimicrobial Prophylaxis to Prevent Urinary Tract Infections after Shockwave Lithotripsy for Urolithiasis: The APPEAL Trial.
Kari A O Tikkinen, Borna Tadayon Najafabadi, Sakineh Hajebrahimi, Farhad Tondroanamag, Arto Mikkola, Saana Horstia, Patrick O Richard, Sara V Tornberg, Mohamed Abdelkareem, Le Mai Tu, Thomas Tailly, Farzin Soleimanzadeh, Hanieh Salehi Pourmehr, Mari Saalasti, Jani Ruotsalainen, Hassan Razvi, Negar Pourjamal, Stephen E Pautler, Niko K Nordlund, Sanna Myrskysalo, Andrey O Morozov, Mohsen Mohammadrahimi, Murilo de Almeida Luz, Samuel Lagabrielle, Pauliina Kuutti, Tuomas P Kilpeläinen, Petrus Järvinen, Alex L E Halme, Alireza Farshi Haghro, Salam A Hussain, Agus Rizal A H Hamid, Dmitry Gorelov, Pramila Gaudel, Nariman Gadzhiev, John Denstedt, Kathrin Bausch, Raed A Azhar; APPEAL Trial Investigators; Khalid Al-Rumaihi, Nourieh D Akbari, Zeenah Abdulmajid, Sameer Parpia, Gordon H Guyatt, Philippe D Violette
Eur Urol. 2025 Dec;88(6):543-551. doi: 10.1016/j.eururo.2025.08.019 FREE ARTICLE
Abstract
Background and objective: Shockwave lithotripsy (SWL) carries a risk of postprocedural infection. Use of antibiotic prophylaxis by clinicians is variable and international guidelines provide conflicting recommendations, reflecting low-certainty evidence. We investigated whether antibiotic prophylaxis reduces bacteriuria and post-SWL urinary tract infections (UTIs).
Methods: APPEAL, an international multicenter, blinded trial, randomized adults undergoing SWL for urolithiasis to a single dose of ciprofloxacin or placebo. The primary outcome was the incidence of a composite of bacteriuria or symptomatic UTI after SWL. Other outcomes included the incidence of pyelonephritis or urosepsis.
Key findings and limitations: Of the 1722 randomized patients, 28 underwent postrandomization exclusions (mainly nonvisualizable stones). Among the analysis population (n = 1694; median age 50 yr; 30% female), 74% had kidney stones and 26% had ureteral stones. Bacteriuria (without symptoms) or symptomatic UTI occurred in 20 patients (2.7%) in the ciprofloxacin arm and 30 (3.9%) in the placebo arm (risk ratio [RR] 0.68, 95% confidence interval [CI] 0.41-1.15). Symptomatic UTI occurred in ten patients (1.3%) in the ciprofloxacin arm and 21 (2.7%) in the placebo arm (RR 0.49, 95% CI 0.19-1.23). No patients in the ciprofloxacin arm and nine (1.2%) in the placebo arm developed pyelonephritis (RR 0.05, 95% CI 0.003-0.93). No patients developed urosepsis and no serious adverse events occurred.
Conclusions and clinical implications: A single dose of ciprofloxacin reduced the risk of post-SWL pyelonephritis, with a modest absolute benefit. The patient importance of this reduction depends on individual preferences in weighing a small absolute reduction in risk against the potential harms and resistance-related implications of antibiotic use. Results from the APPEAL trial will inform global practice and support evidence-based decision-making for patients undergoing SWL.
Comment Peter Alken
I spent a lot of thoughts on this paper and two of my four questions were: 1) Is the question addressed in the study important and urgent? 2) Or - if not - what made the study important?
I have read this free access paper and reference 13 which was not readily available for me on PubMed. However, I could access it as a preprint (1): [medRxiv preprint doi: https://doi.org/10.1101/2025.03.09.25323609; this version posted March 10, 2025. The copyright holder for this preprint (which was not certified by peer review) is the author/funder, who has granted medRxiv a license to display the preprint in perpetuity. It is made available under a CC-BY-NC-ND 4.0 International license.]. There it says “This trial commenced recruitment in London (Ontario, Canada) in 2013 as a single-centre trial. Recruitment challenges are common in randomized surgical trials: slow recruitment is the most common reason for discontinuation. This trial faced similar challenges.” “We expanded the trial globally and renamed as APPEAL.”
In the present paper it says: “Following a single-center trial in London (Canada) from January 2013 to December 2015, we expanded recruitment between September 2016 and July 2022 [16], ultimately enrolling 1722 patients from 12 centers across nine countries.”
In the “Advancing Practice” section added to the present paper in European Urology it says “Given the modest absolute risk reduction, the decision to use prophylaxis should be individualised, incorporating patient values and preferences. Our findings call for guideline updates and standardised prophylaxis practices of shared decision-making. Research should focus on identifying the high-risk subgroups that could benefit the most.”
I agree and do not see a real advancement of knowledge concerning the topic by this paper because I think that this is already done in real life today - with the usual exceptions. So, what made the study so important?
In the preprint paper the use of ciprofloxacin is discussed: ”In 2019, the European Commission issued a legally binding decision restricting the use of quinolone antibiotics, including ciprofloxacin, across all EU countries, primarily because of adverse effects associated with prolonged use [30] (2). This decision has influenced opinions on suitable antibiotic prophylaxis strategies, particularly in the context of urinary procedures for which quinolones were commonly used. Despite this, we believed that continuation of recruitment for APPEAL was ethical because i) evidence has shown that the adverse effects of quinolones are overwhelmingly associated with long-term use, and ii) ciprofloxacin remains one of the prophylactic antibiotics most often used in urology.”
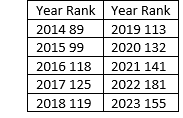
The 2019 EMA-report was based on an EMA hearing on fluoroquinolone and quinolone antibiotics in June 2018. In 2018 the rumours on problems with these compounds were on the market since some time. Some of these issues are dealt with in the references attached (4,5). The overall US Drug Usage Statistics, 2014 – 2023 (Table 1) (3) shows that ciprofloxacin, the previous blockbuster is not doing so well during the years.
Table 1
Ciprofloxacin Usage Statistics in the US
Modified from https://clincalc.com/DrugStats/Drugs/Ciprofloxacin
"Rank" refers to the frequency that a given medication is prescribed within a calendar year
compared to all other medications. A rank of "4" would indicate that the medication was the fourth
most commonly prescribed medication.”
The third question was 3) How was assured that the drug was taken and that it was the identical, proper compound in the different countries? And the fourth question is 4) Will this paper change the antibiotic use in ESWL?
The actually presented AUA guideline on urolithiasis treatment states:” For adult patients with kidney or ureteral stones undergoing SWL, clinicians may omit preoperative prophylactic antibiotics. (Conditional Recommendation; Evidence Level: Grade B)
SWL is a non-invasive procedure that does not require routine use of preoperative antibiotic prophylaxis in the absence of preoperative evidence of urinary tract infection (UTI)”(6).
I selected it as “Reviewer’s Choice” because I think the attentive reader might have more than four questions and that the work offers some insights into research and publication practices.
1 www.medrxiv.org/content/10.1101/2025.03.09.25323609v1.full.pdf+html
2 European Medicines Agency. Quinolone- and fluoroquinolone containing medicinal products.
Amsterdam, The Netherlands: European Medicines Agency; 2019.
3 https://clincalc.com/DrugStats/Drugs/Ciprofloxacin
4 Omrani MA, Tsobo CT, Chalabianloo N, Ahmadi F, Abdullah SS, Muanda FT. Fluoroquinolones and risk of nightmares: A literature review and disproportionality analysis using individual case safety reports from Food and Drug Administration Adverse Event Reporting System database. J Psychopharmacol. 2025 Aug;39(8):782-789. doi: 10.1177/02698811251344684. Epub 2025 Jun 27. PMID: 40576985; PMCID: PMC12287549.
5 Yang L, Chen C, Ding L, Lu T, Li X, Xiao J. Suicidal thoughts and behaviors associated with fluoroquinolone antibiotics: a real-world pharmacovigilance analysis. Front Pharmacol. 2025 Apr 25;16:1556159. doi: 10.3389/fphar.2025.1556159. PMID: 40351425; PMCID: PMC12062094.
6 Pearle MS., et al.
Surgical Management of Kidney and Ureteral Stones: AUA Guideline (2026) Part II: Evaluation and Treatment of Patients with Kidney and/or Ureteral Stones. The Journal of Urology Publish Ahead of Print DOI: 10.1097/JU.0000000000004843
Peter Alken


Comments